Compounds containing an all-metal antiaromatic ring isolated for the first time
An international research team has prepared a set of lanthanide antimony clusters that represent the first isolable compounds containing an all-metal antiaromatic ring. The achievement continues to expand the concept of aromaticity beyond its humble beginnings 150 years ago.
Researchers including Xue Min and Zhong-Ming Sun of Changchun Institute of Applied Chemistry and Ivan A. Popov and Alexander I. Boldyrev of Utah State University created a series of anions, [Ln(Sb4)3]3–, where Ln is La, Y, Ho, Er, or Lu. They made the anions by treating lanthanide benzyl complexes with the Zintl cluster complex K5Sb4 in pyridine solvent and then isolating the anions as potassium cryptand salts.
On the basis of X-ray crystal structures and computational bonding analysis, the team says the rhombic Sb4 rings that serve as ligands to the lanthanide metals are antiaromatic (Angew. Chem. Int. Ed. 2016, DOI: 10.1002/anie.201600706).
The concept of antiaromaticity has a storied history. In 1865, German chemist August Kekulé proposed the concept of aromaticity to explain the unusual properties of benzene, a planar carbon ring that exhibits high stability and low reactivity. In 1931, German chemist Erich Hückel added to the definition that aromatic compounds have a delocalized 4n + 2 π-electron system. In 1965, on the centennial of Kekulé’s proposal, Columbia University’s Ronald Breslow proposed the idea of antiaromaticity—the antonym of aromaticity—to characterize planar carbon rings with a 4n π-electron system that exhibit low stability and high reactivity.
Aromaticity and antiaromaticity were originally thought to be purely the domain of organic chemistry. But during the past 20 years, chemists have shown that this organic boundary is flexible. In 1995, Gregory H. Robinson and coworkers of the University of Georgia isolated the sodium salt of a phenyl-substituted Ga3 ring with two π-electrons, introducing the concept of metalloaromaticity.
In 2003, Boldyrev’s group in collaboration with Lai-Sheng Wang, now at Brown University, followed suit by reporting Li3Al4–, which includes an antiaromatic Al44– ring containing four π electrons. However, the gaseous molecule was created in a laser-based experiment and couldn’t be trapped in a condensed state.
With the [Ln(Sb4)3]3– series, chemists now have the first examples of isolable inorganic antiaromatic compounds. As a key feature, each Sb4 ring stabilized by the lanthanide metal has four delocalized π electrons. The Sb4 unit is analogous to cyclobutadiene, Boldyrev says, which is the quintessential antiaromatic organic compound.
“Antiaromaticity in these all-metal systems is very nice,” Breslow tells C&EN. “It is gratifying to see that our proposal, which was quite unexpected when we first made it for organic systems, has such generality.”
Further advances of aromaticity and antiaromaticity into metal territory will be valuable for understanding the properties of metal clusters, bulk metals, and alloys, Boldyrev and Sun add, which could be handy for making thin-film electronic materials.
“From a conceptual perspective, this is another example of the concept of aromaticity—in this case antiaromaticity or antimetalloaromaticity—being extended beyond the realm of carbon,” Robinson says. “More important, taking all of this work into consideration, aromaticity and metalloaromaticity seem to be foundational principles throughout the whole of chemistry.” (C&EN news http://cen.acs.org/articles/94/i16/Cyclic-antimony-complexes-take-aromaticity.html)
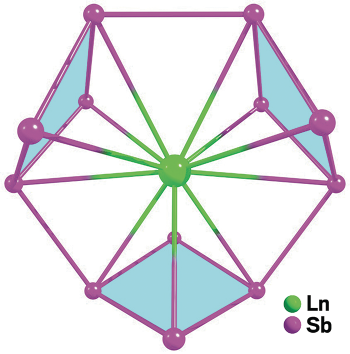
In the [Ln(Sb4)3]3– compounds, a lanthanide(III) cation coordinates three antiaromatic Sb4 rings.
(Image by Ivan Popov)
Contact:
SUN Zhong ming
Email: szm@ciac.ac.cn
State Key Laboratory of Rare Earth Resources Utilization, Changchun Institute of Applied Chemistry
